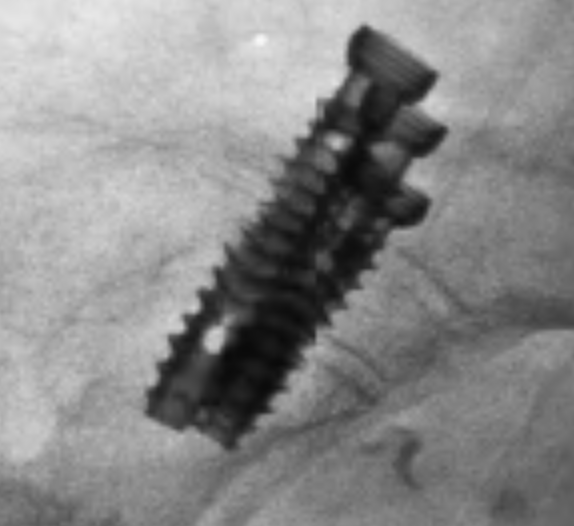
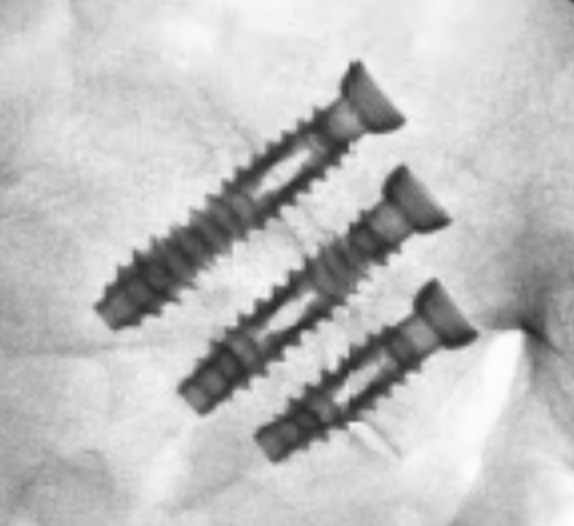
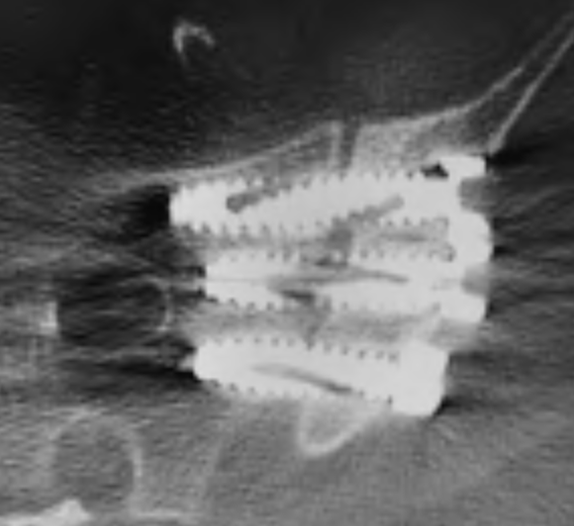
SI-Cure Implant
The SI-Cure Implant was conceived and designed to optimize the treatment of patients with SI joint pain by providing compression, fixation and fusion. We have more than 5-years of clinical experience using a straightforward and clinically proven lateral technique. By focusing on fusion, the SI-Cure Implant offers patients the potential for great outcomes.